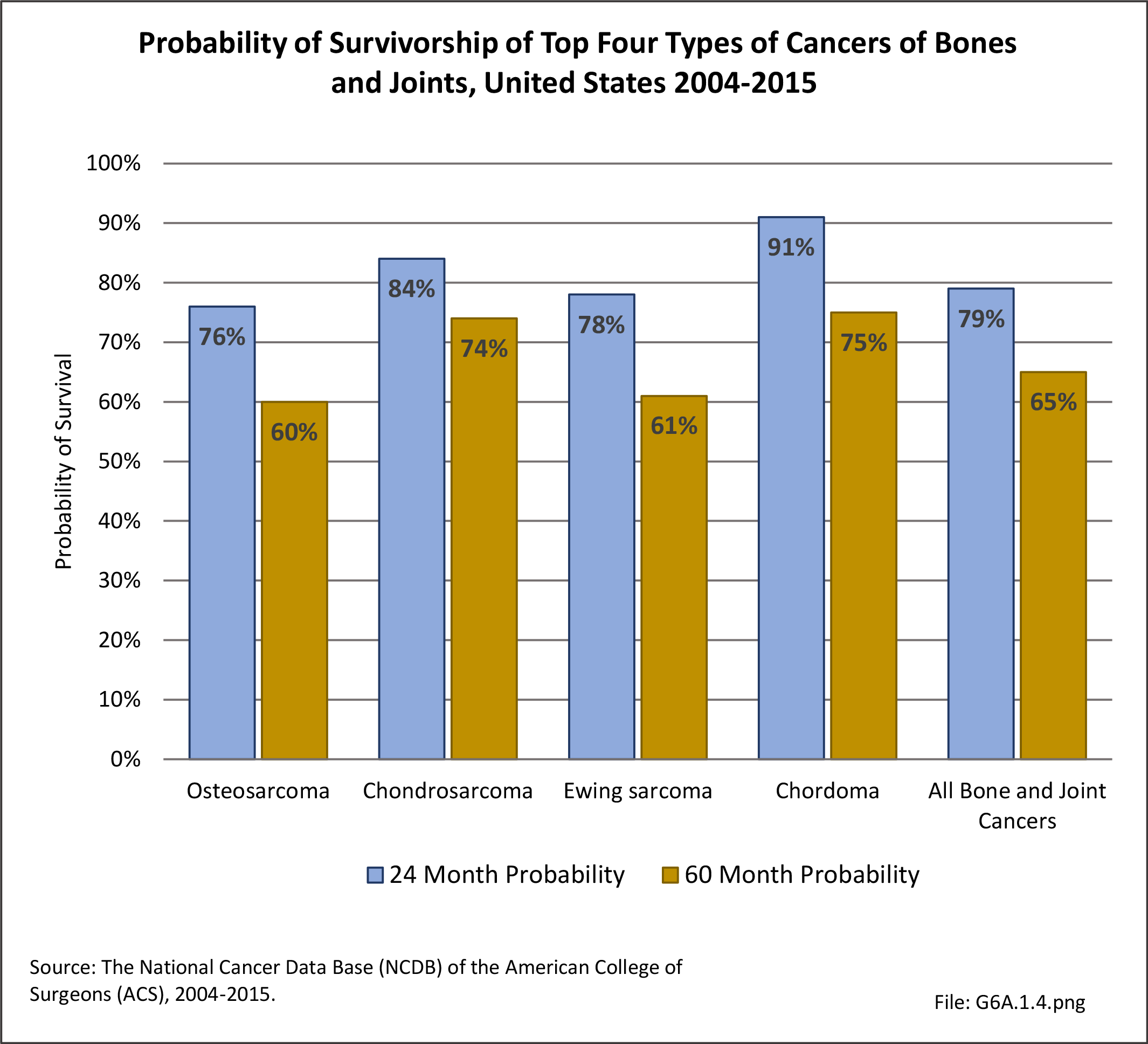
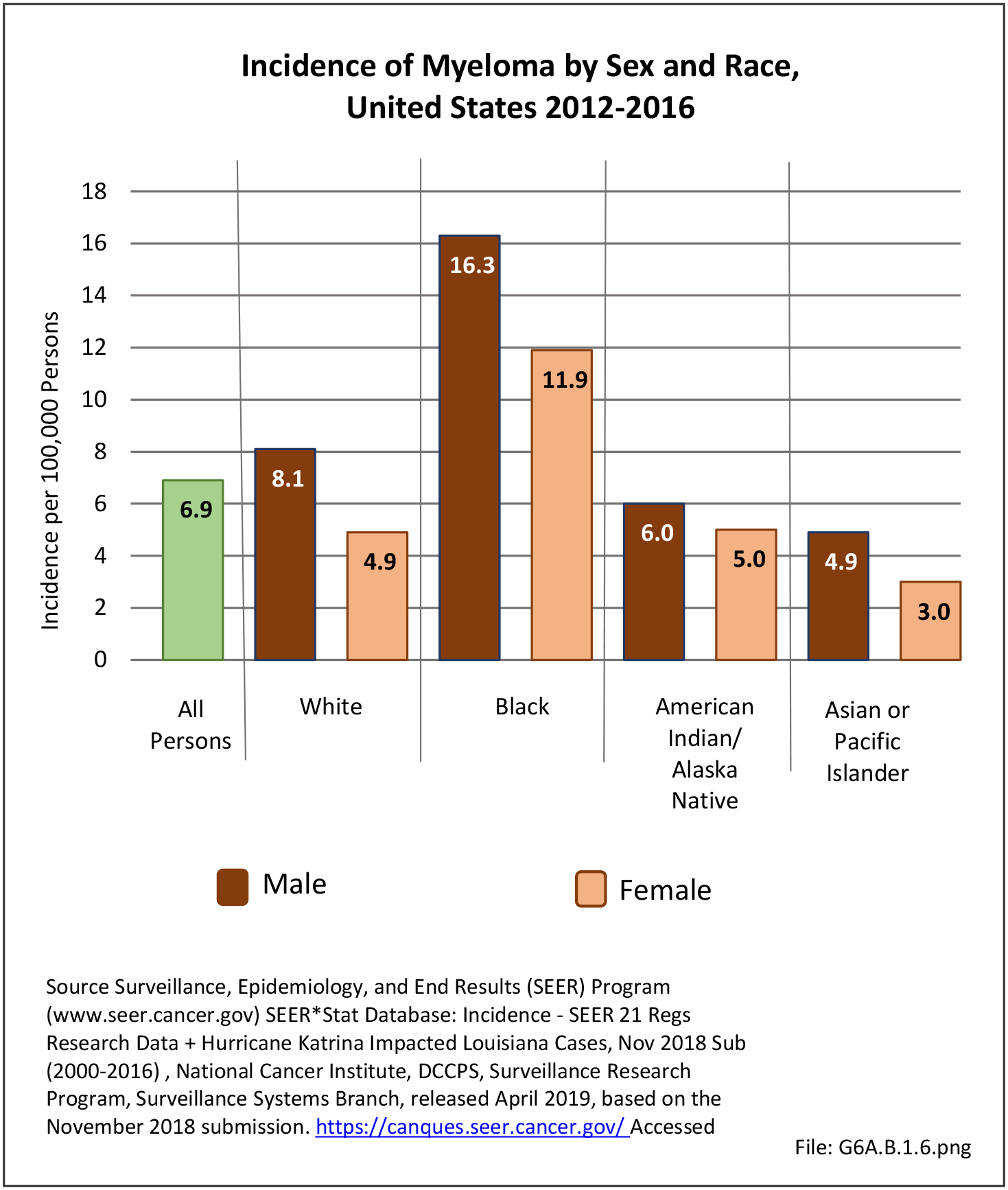
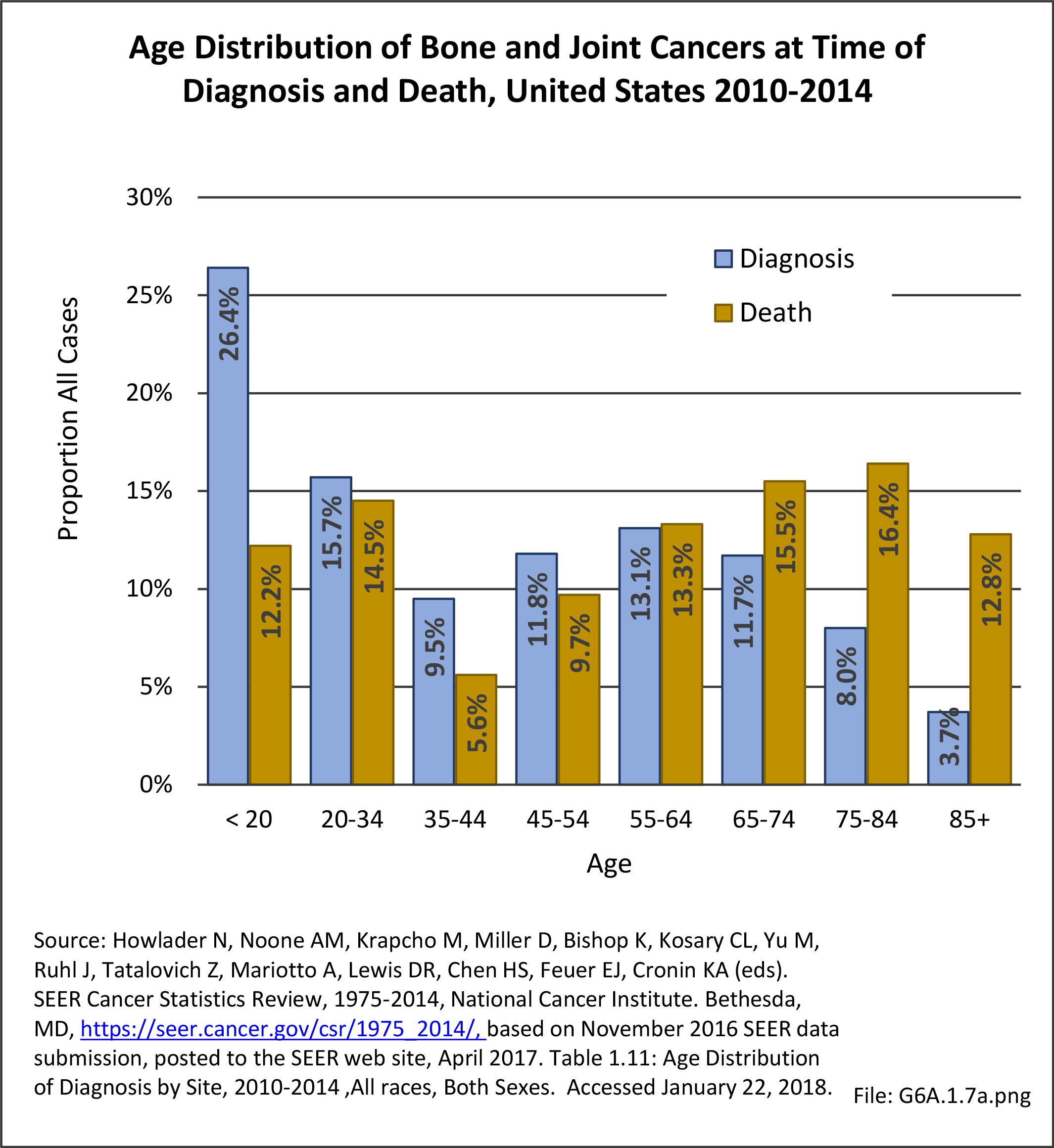
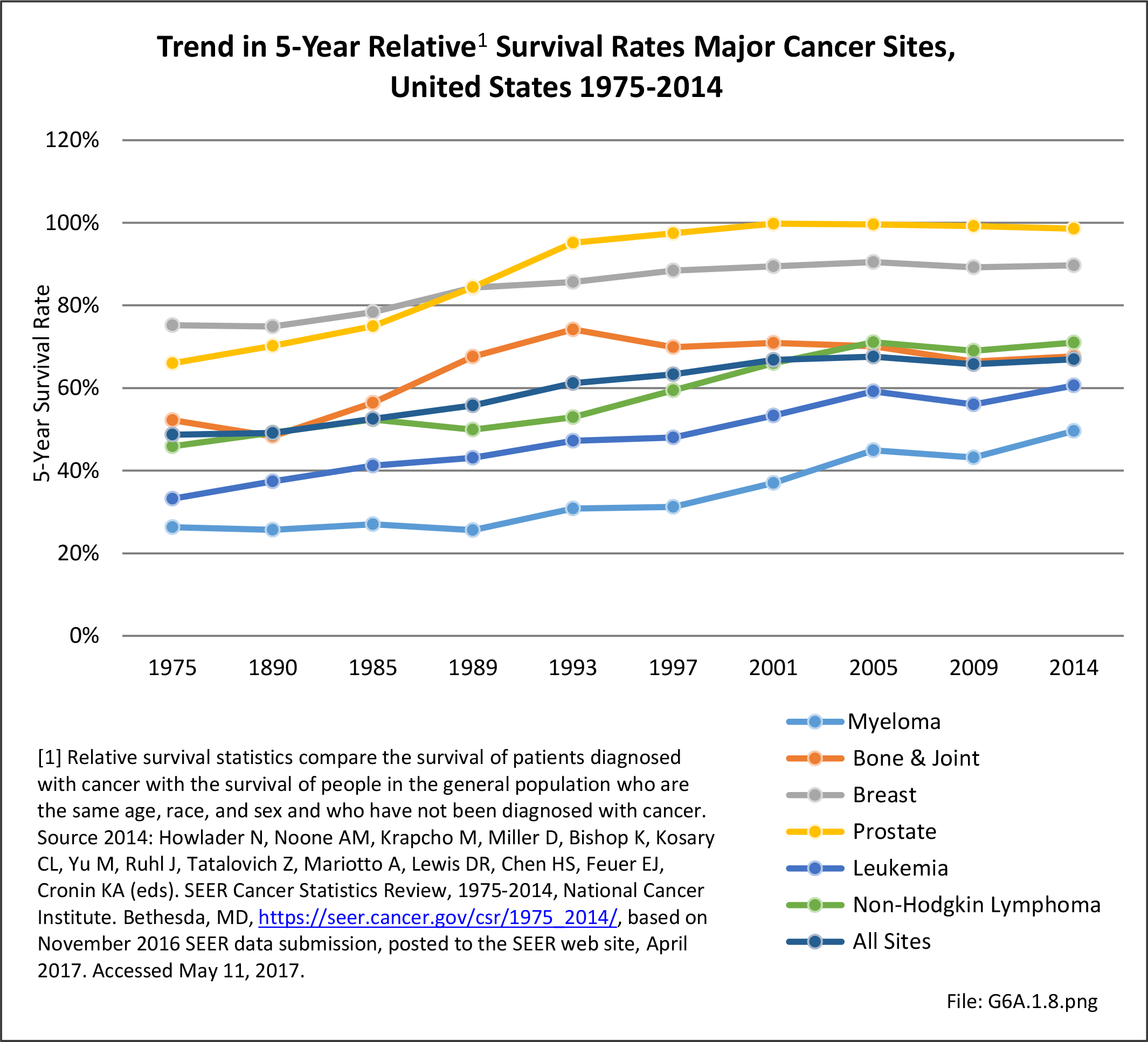
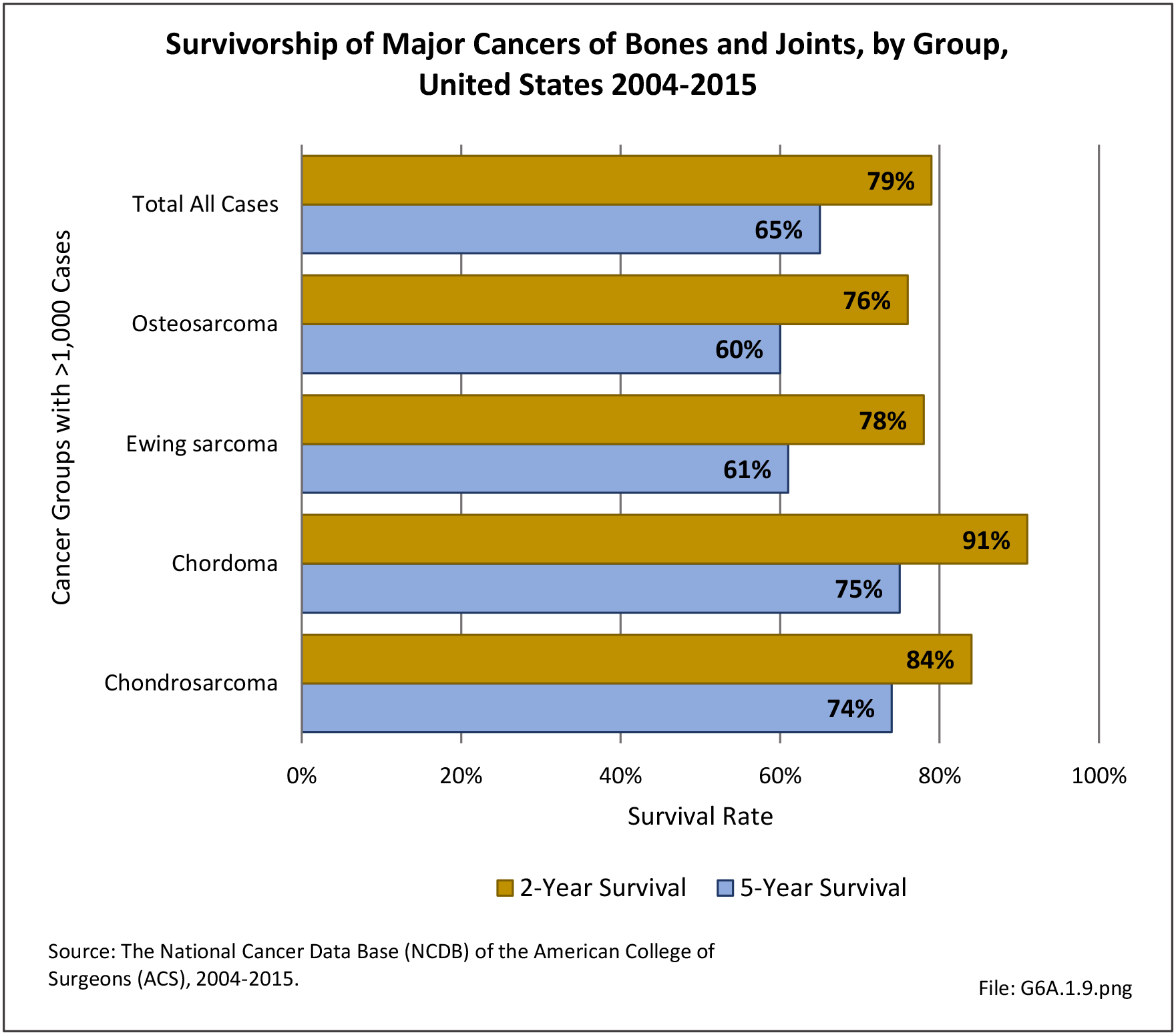
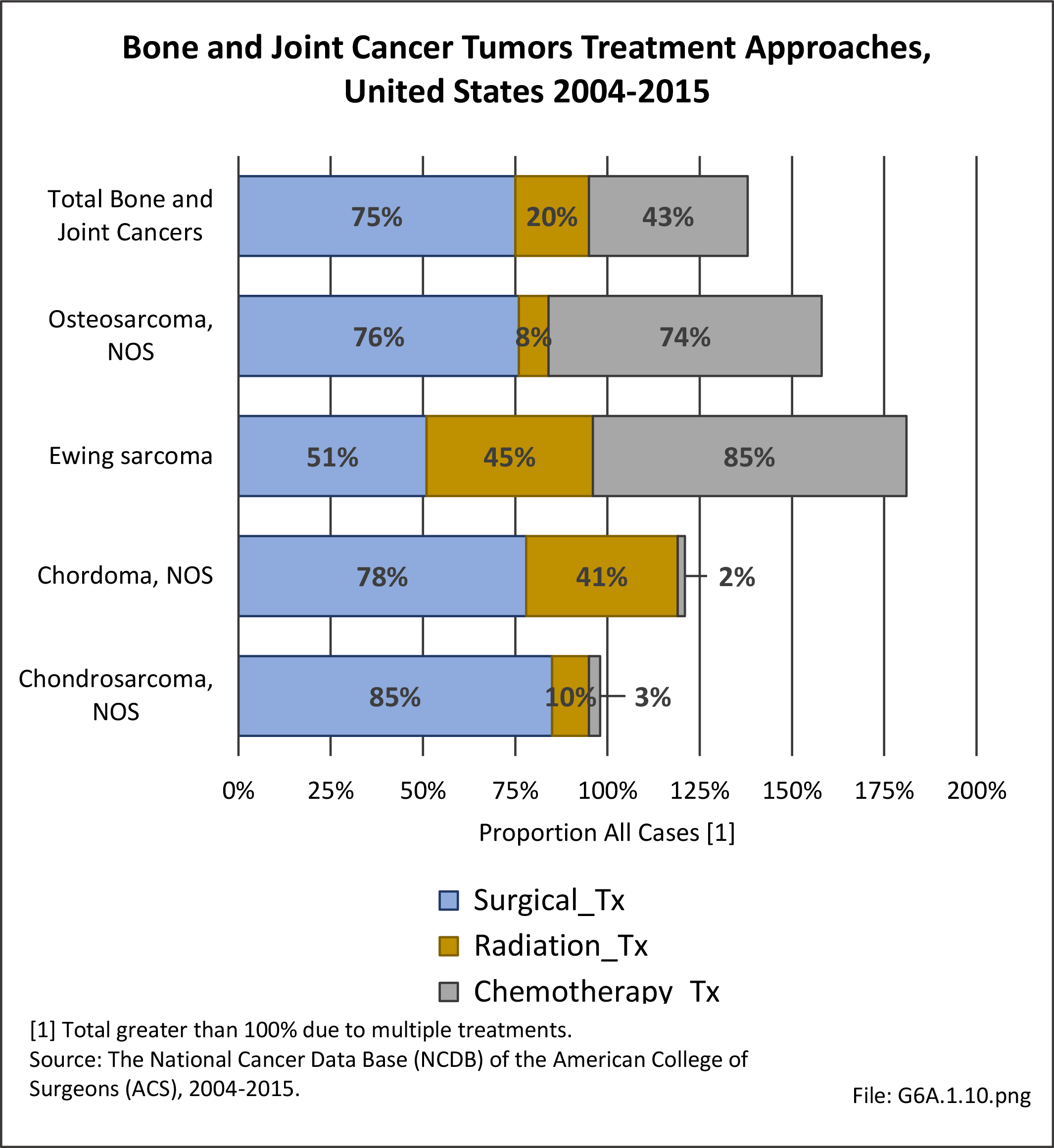
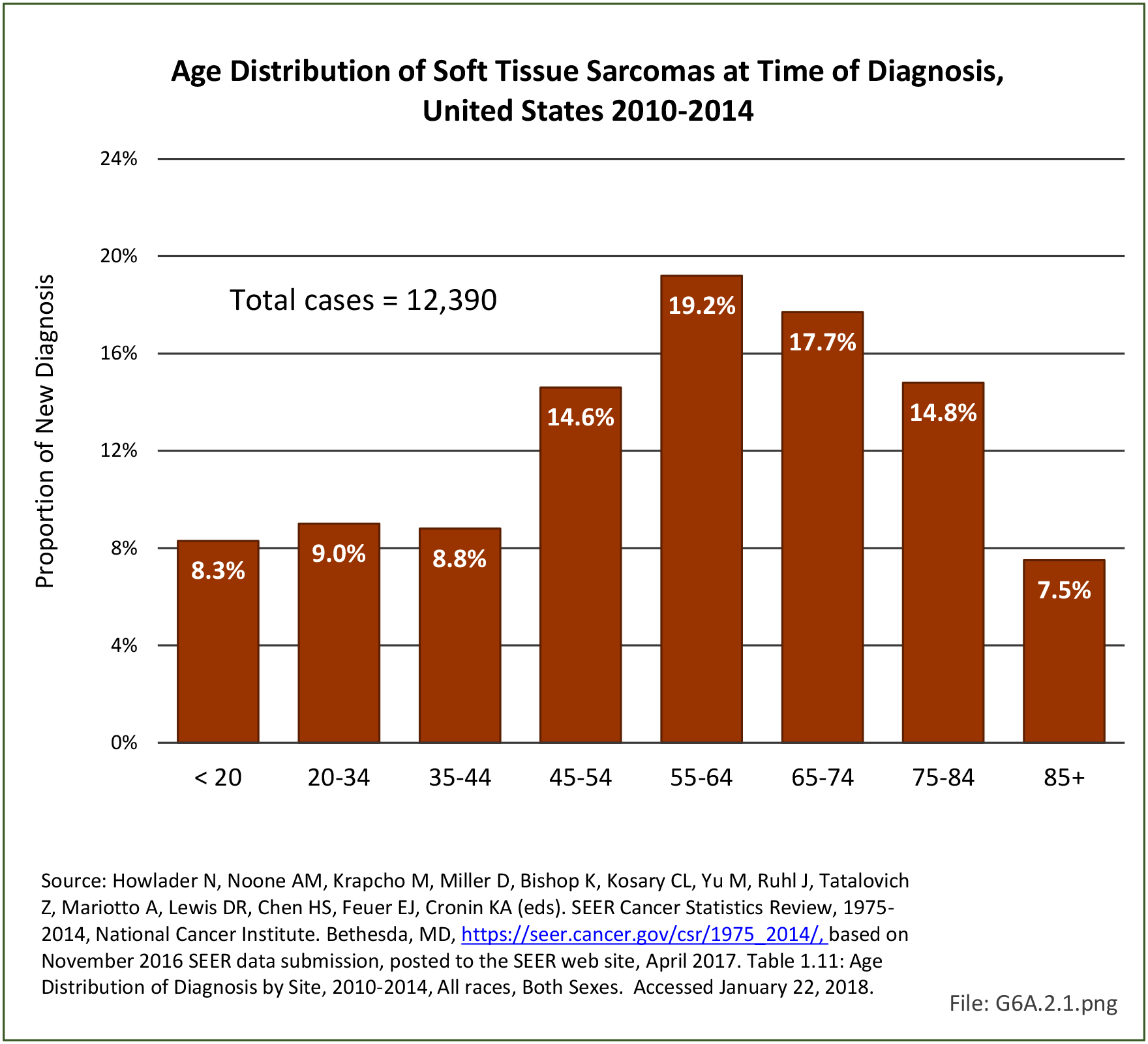
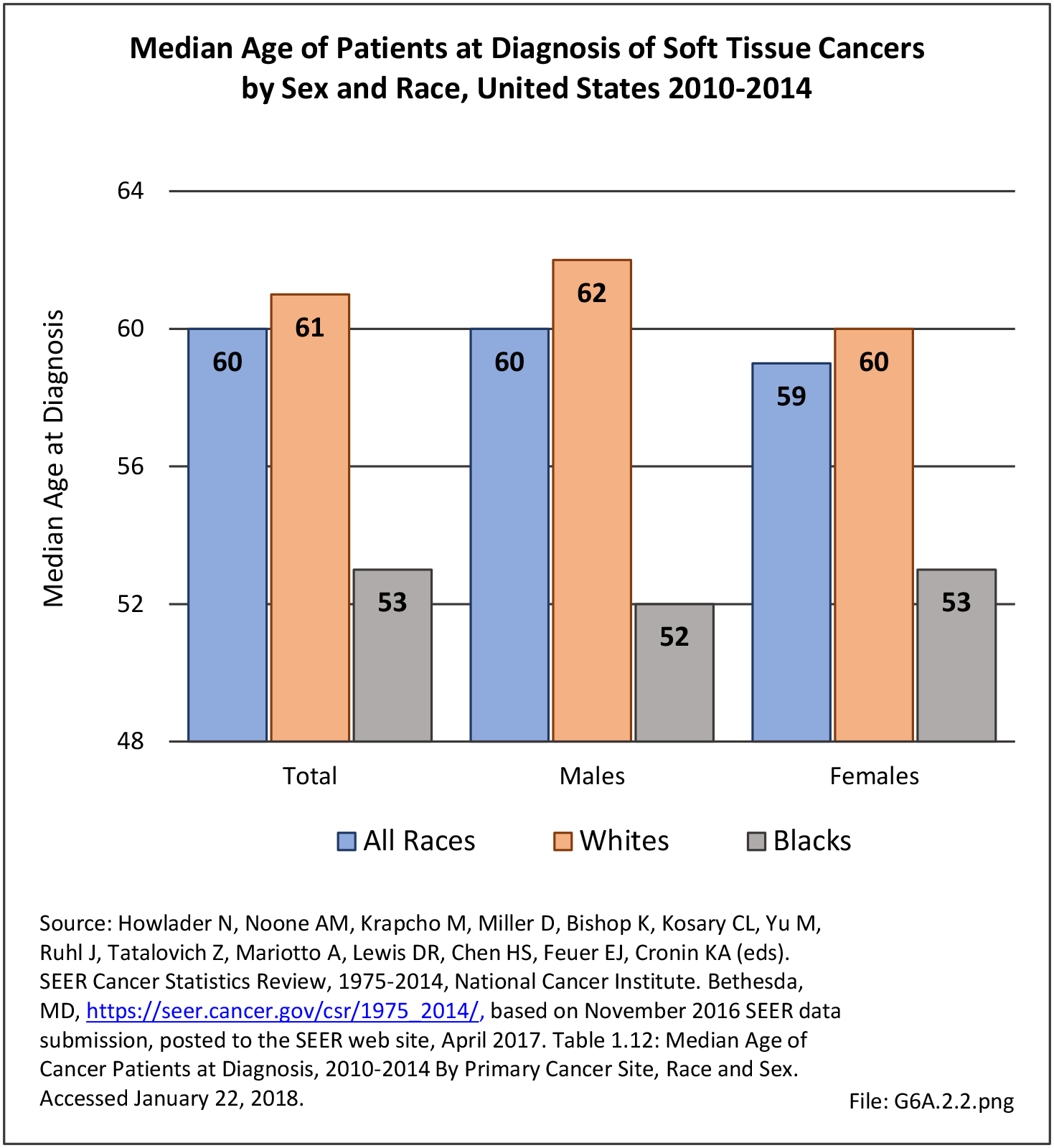
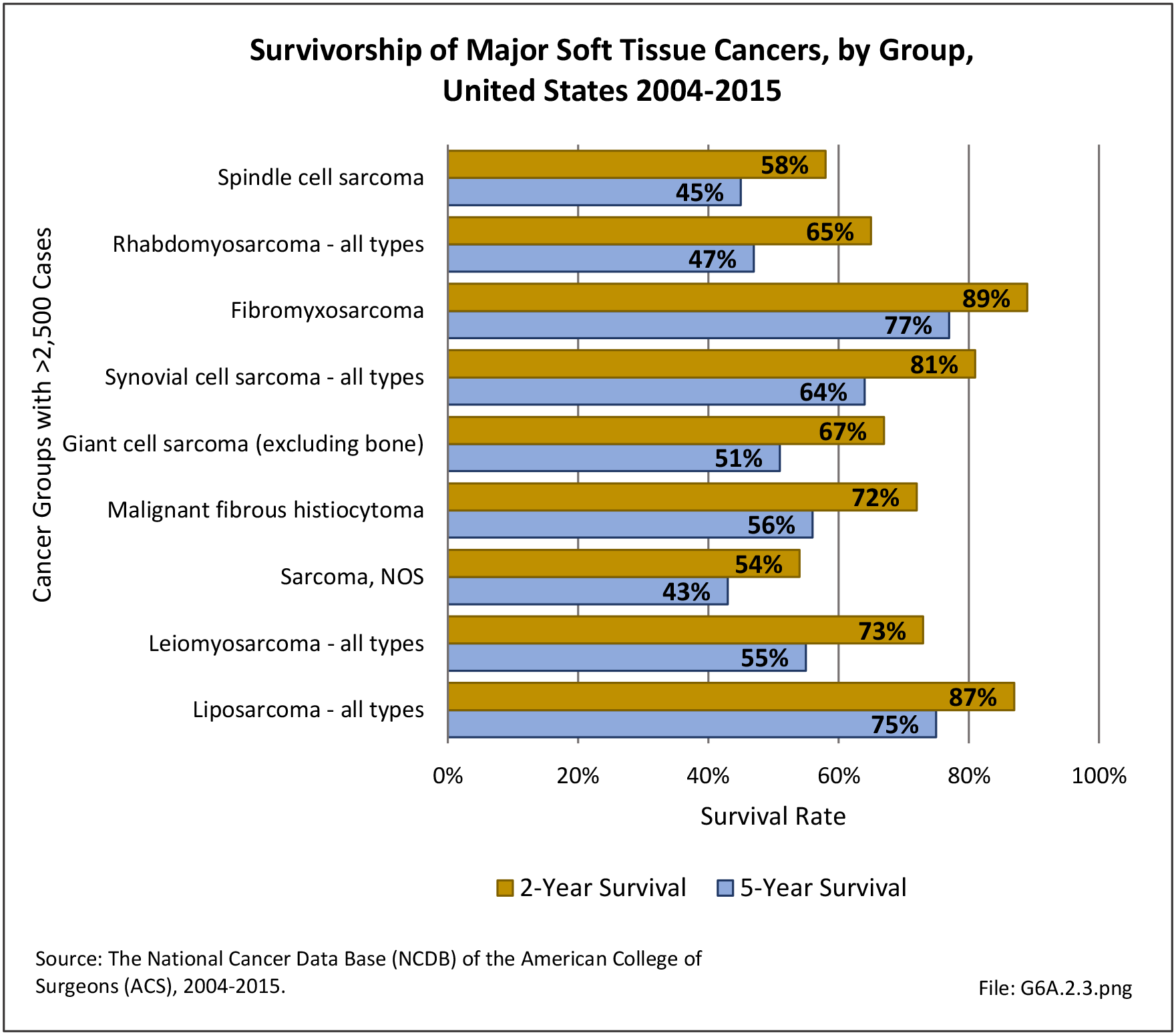
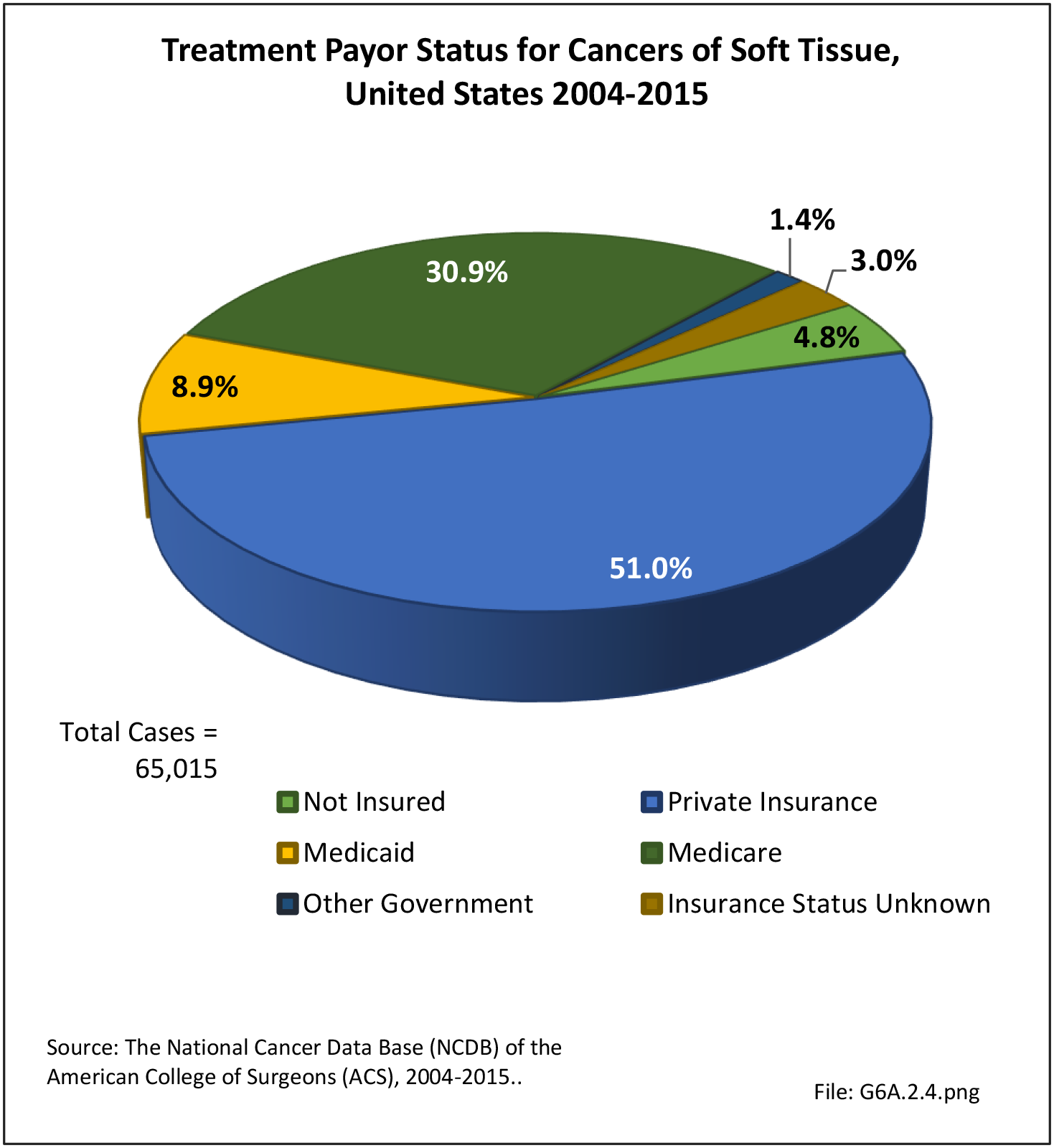
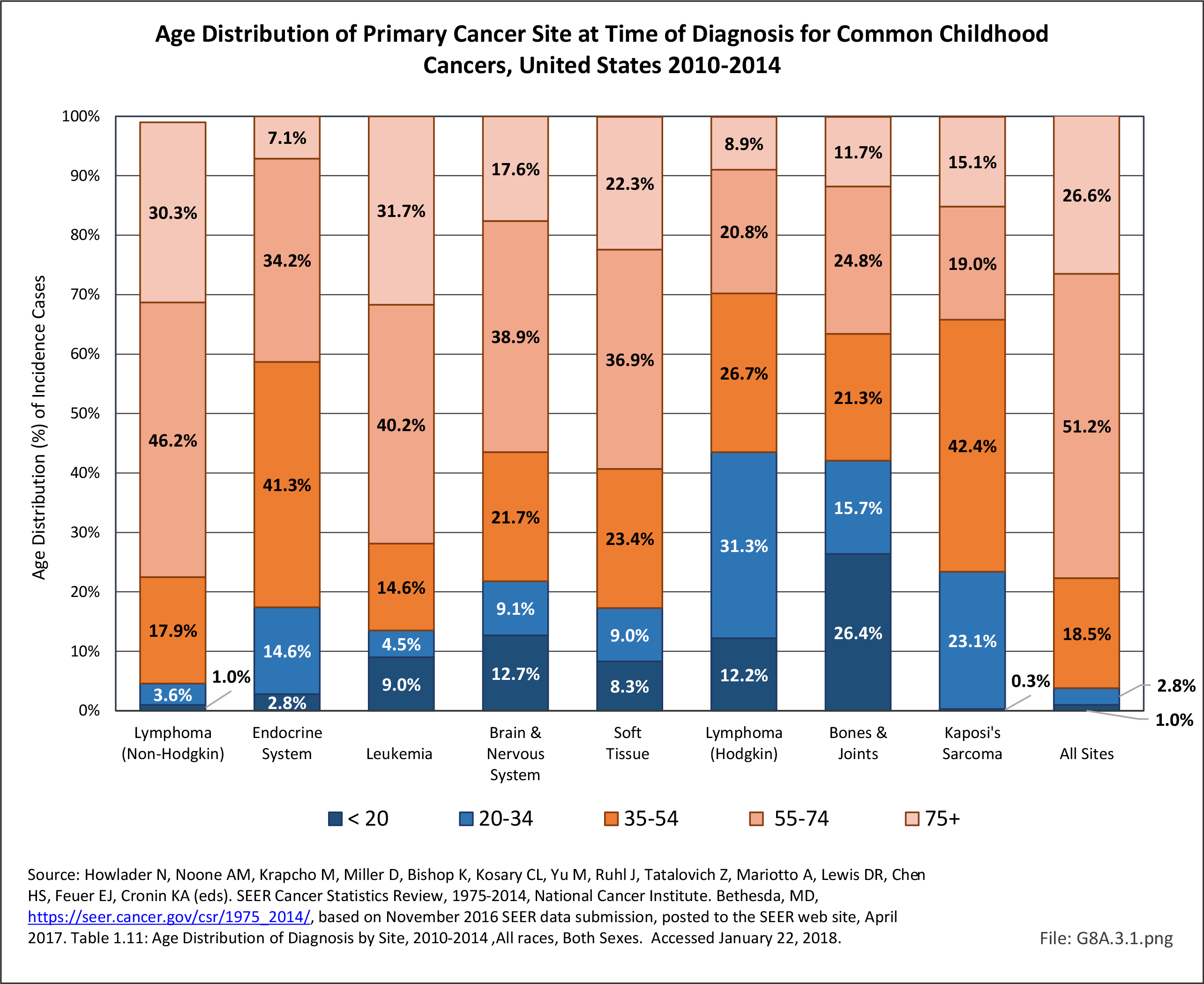
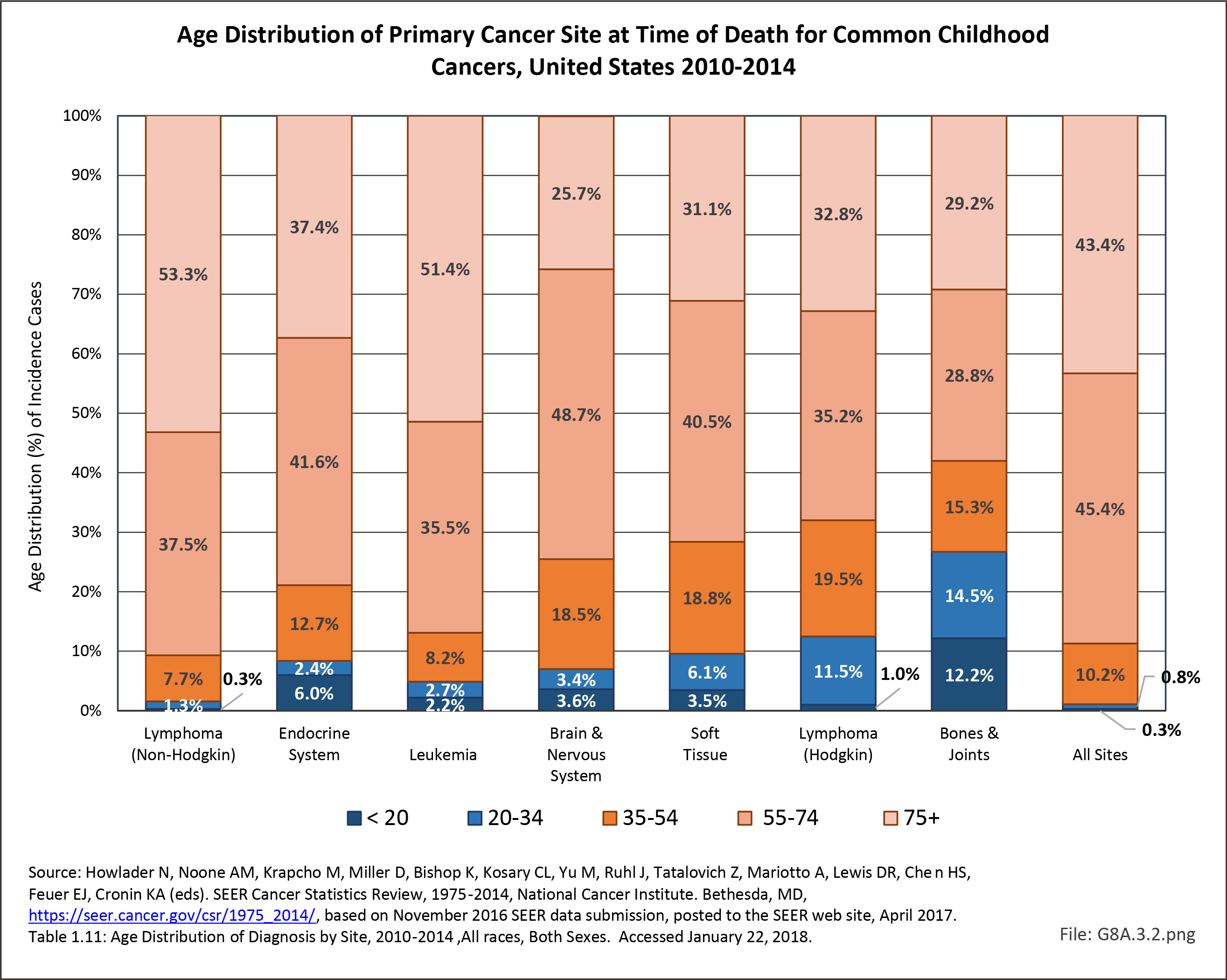
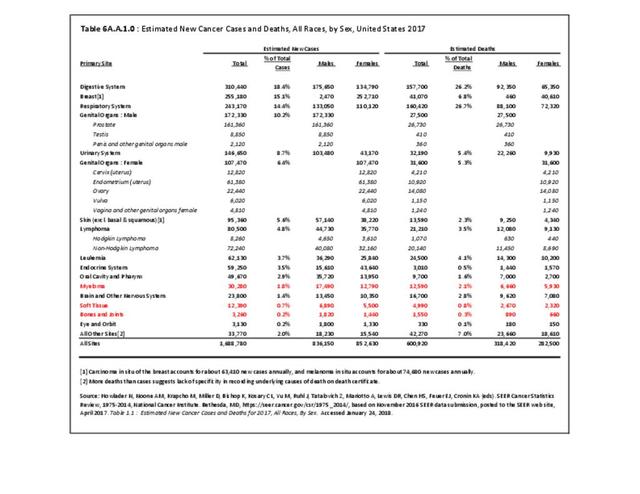
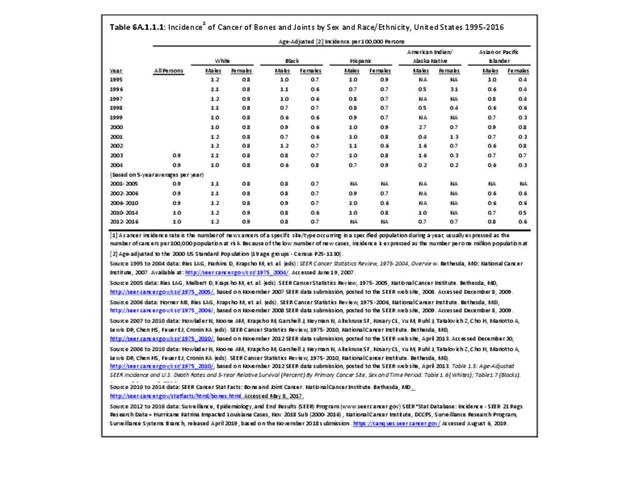
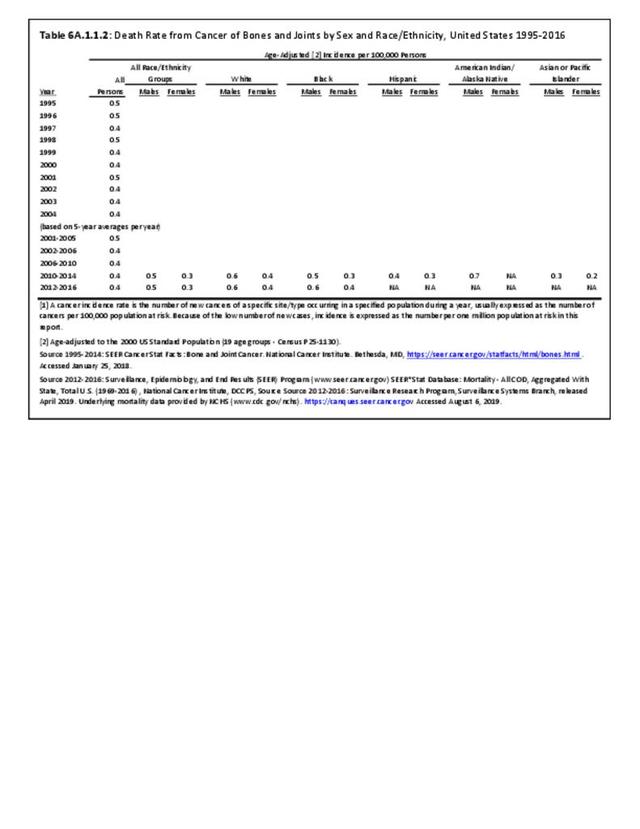
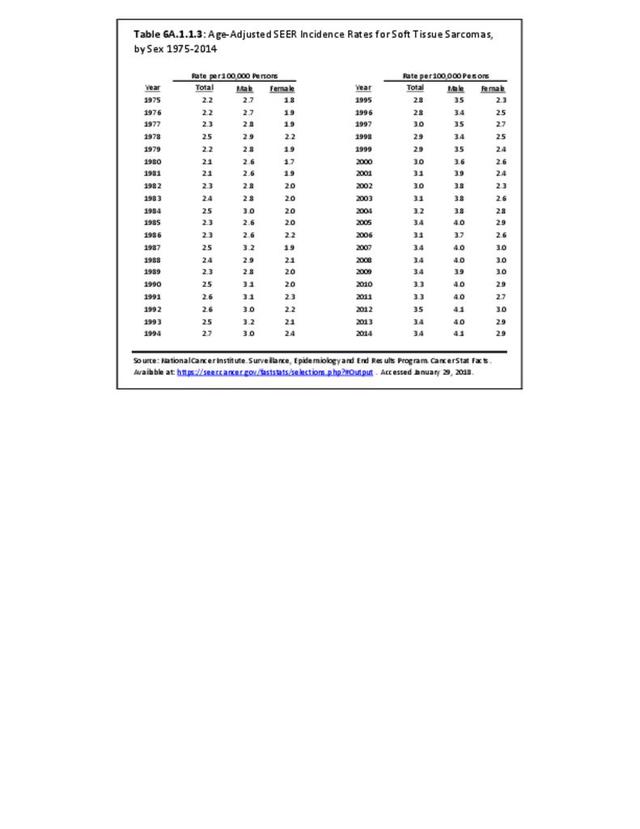
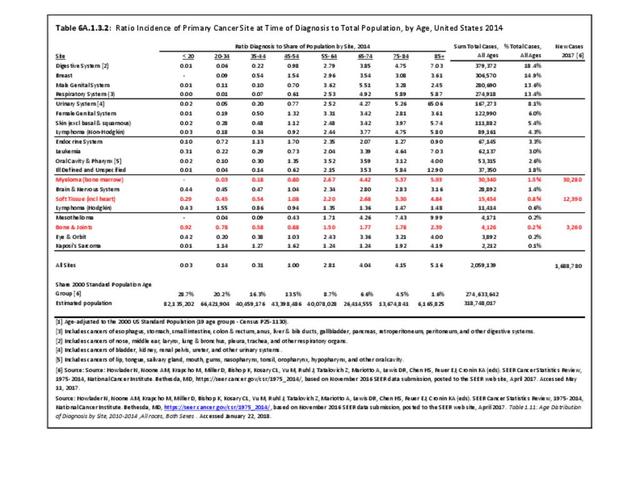
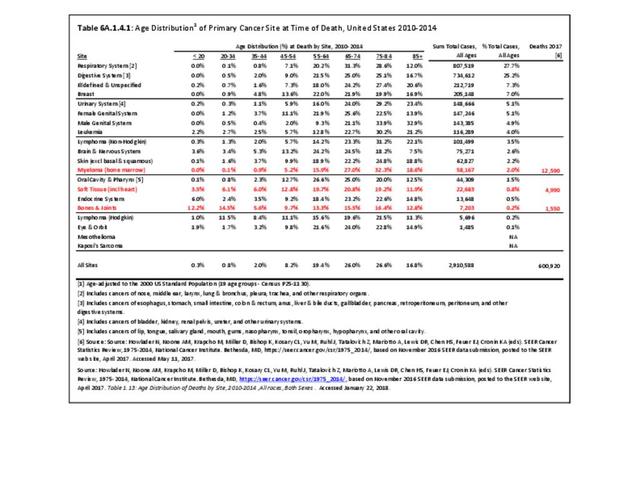
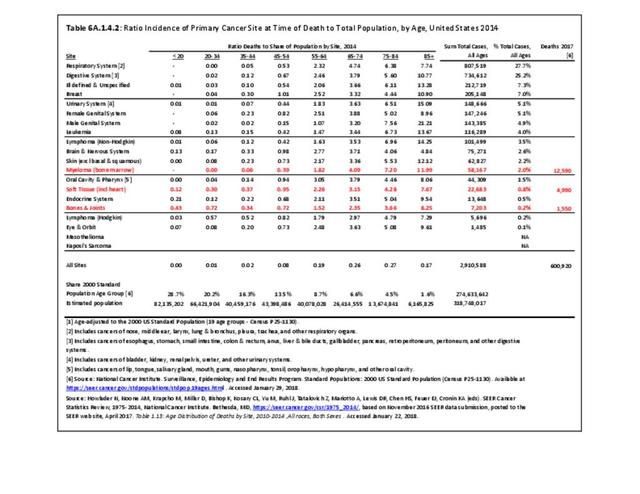
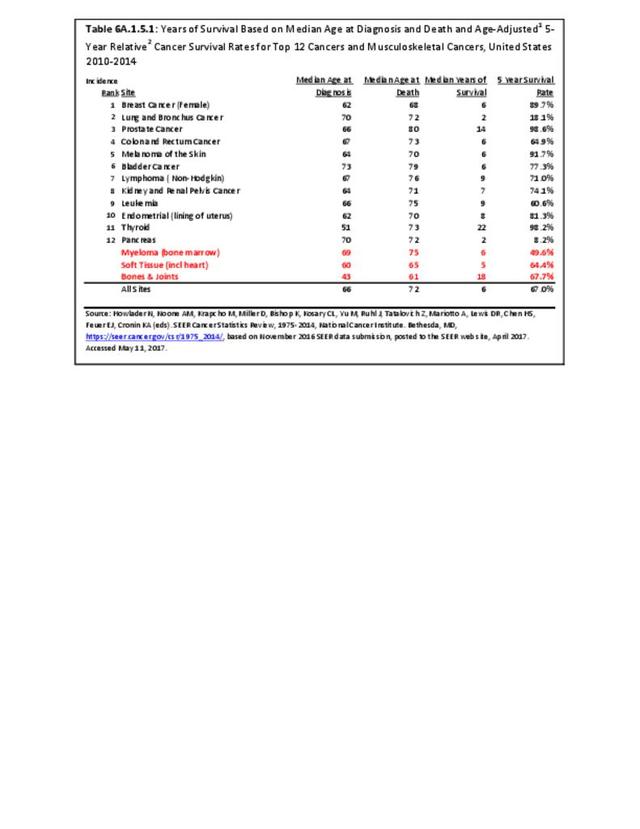
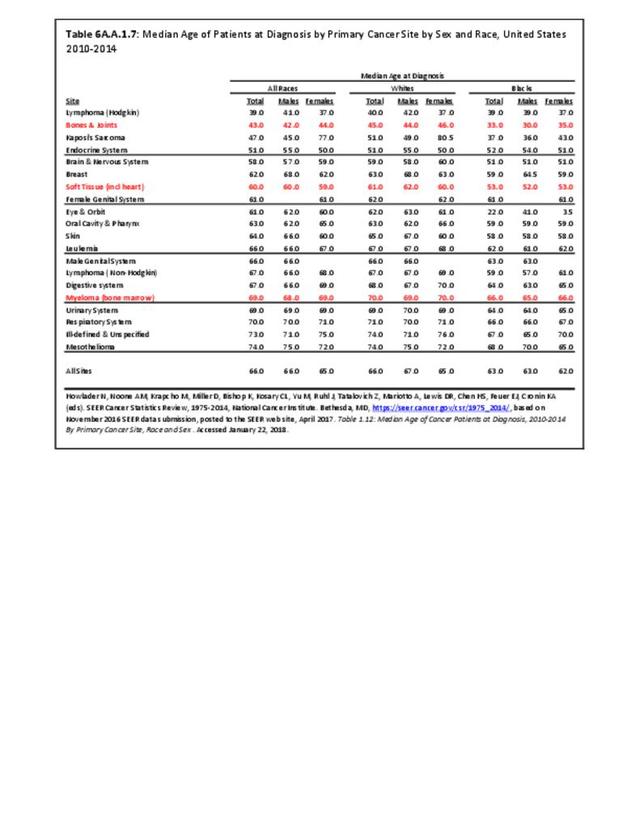
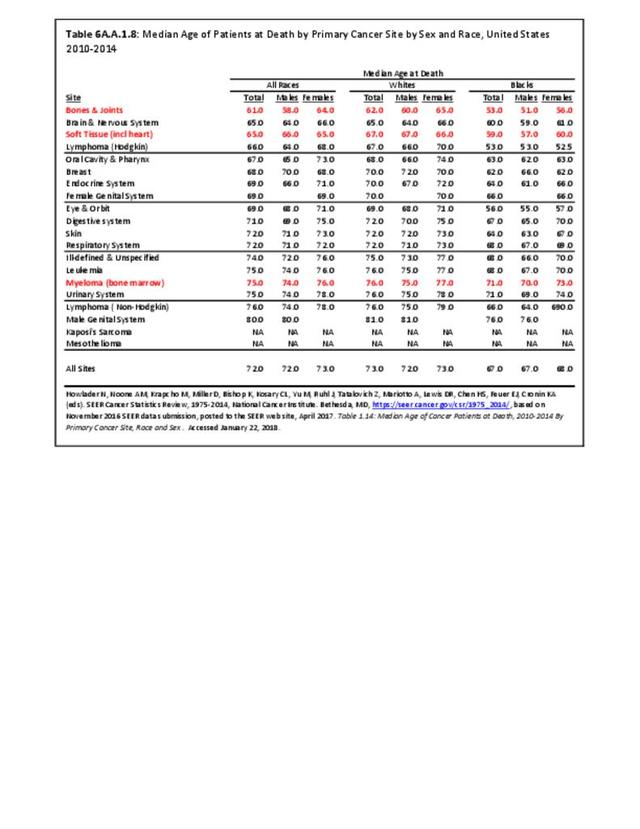
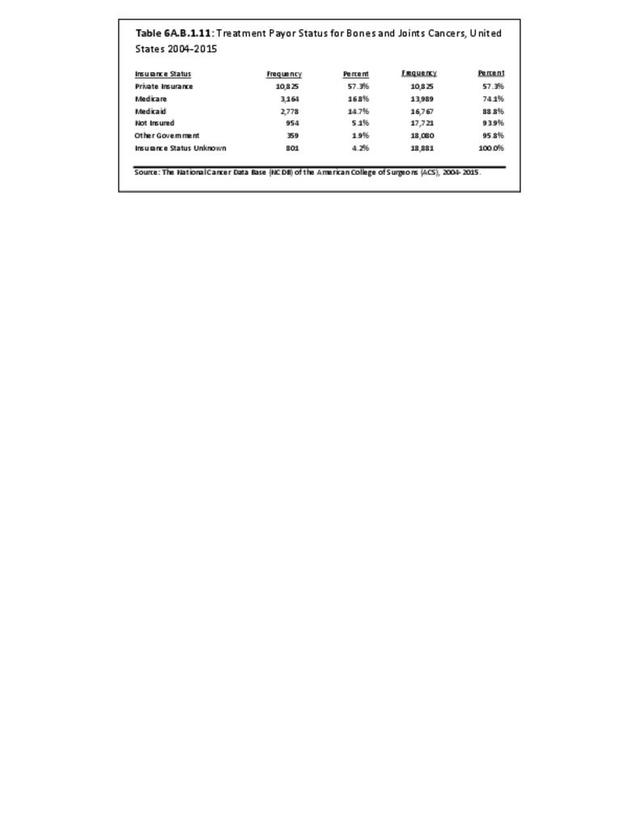
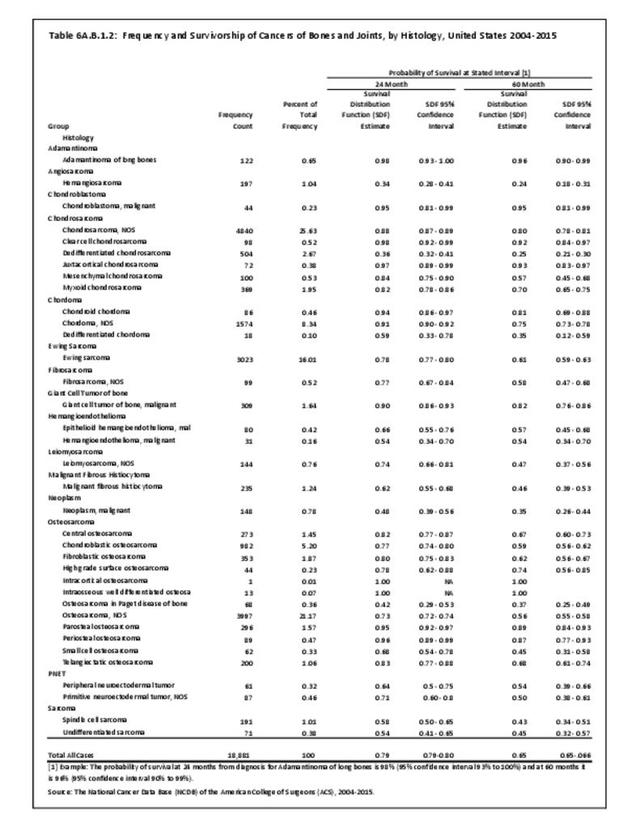
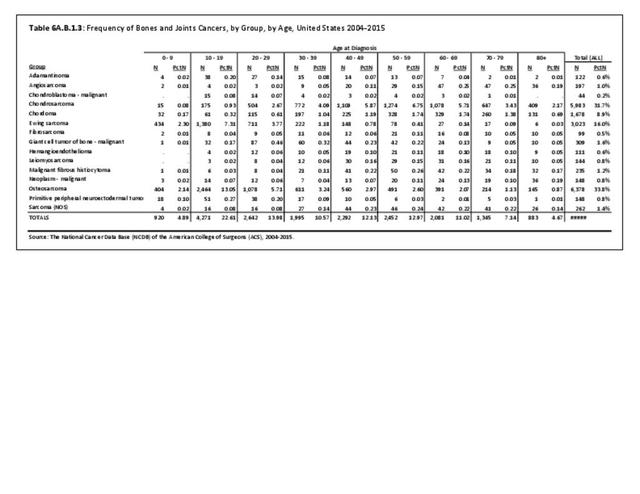
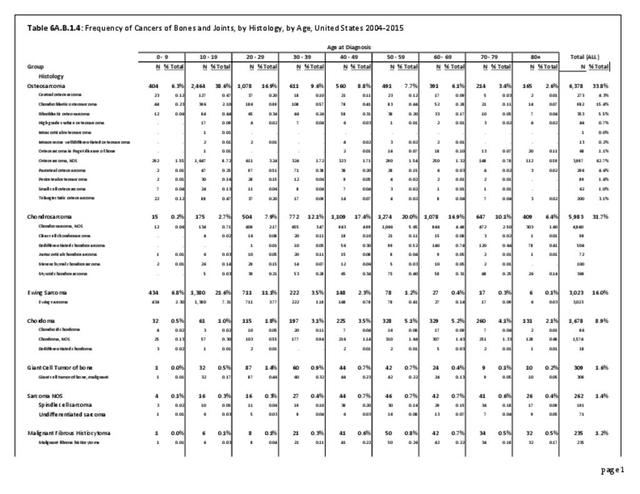
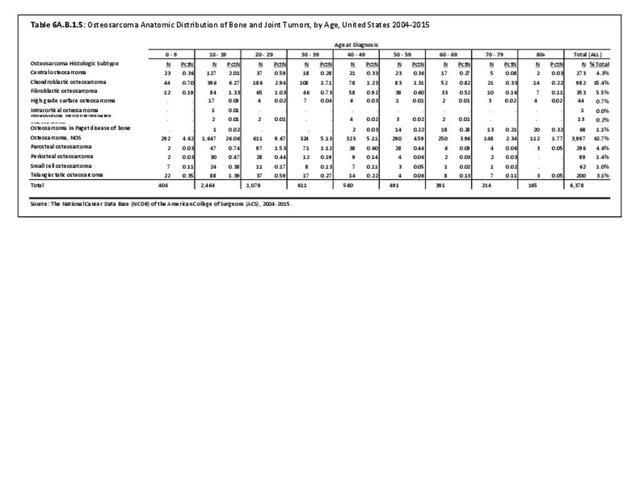
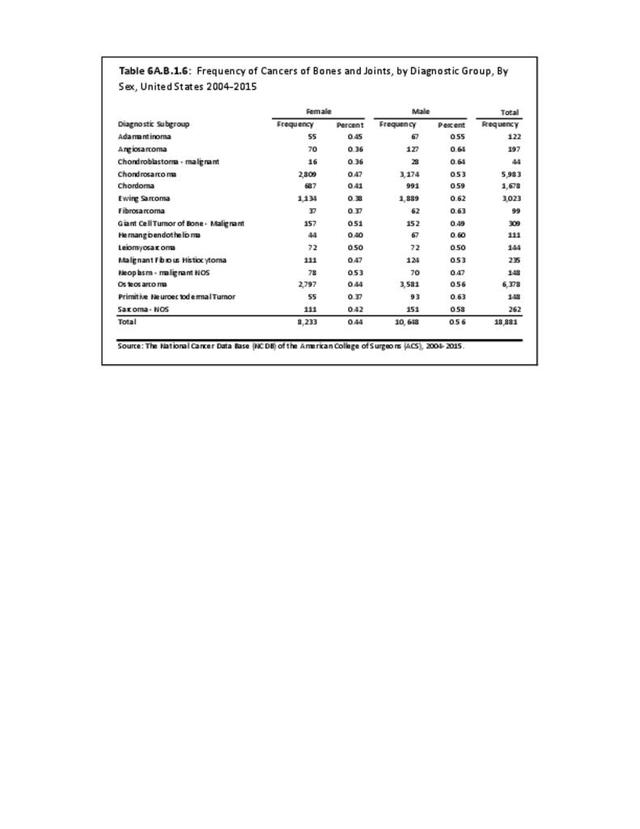
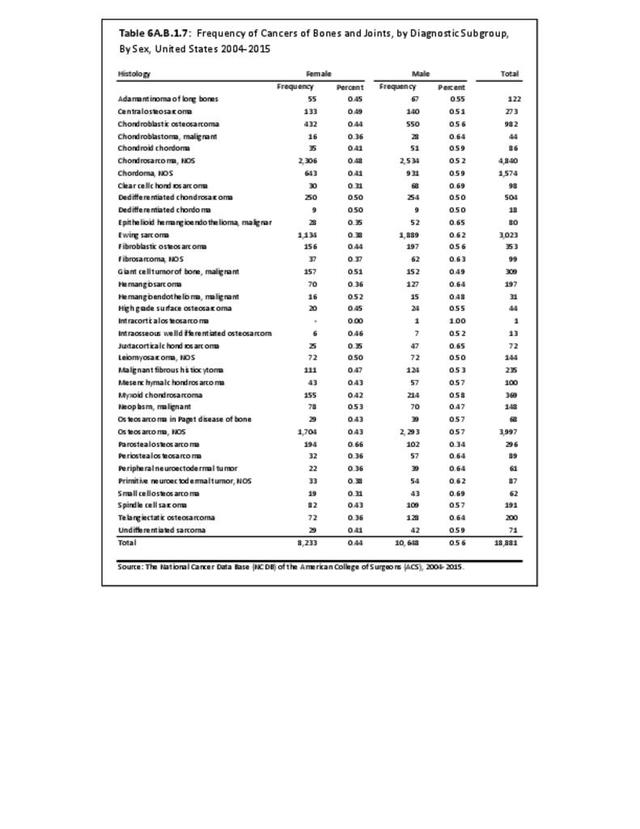
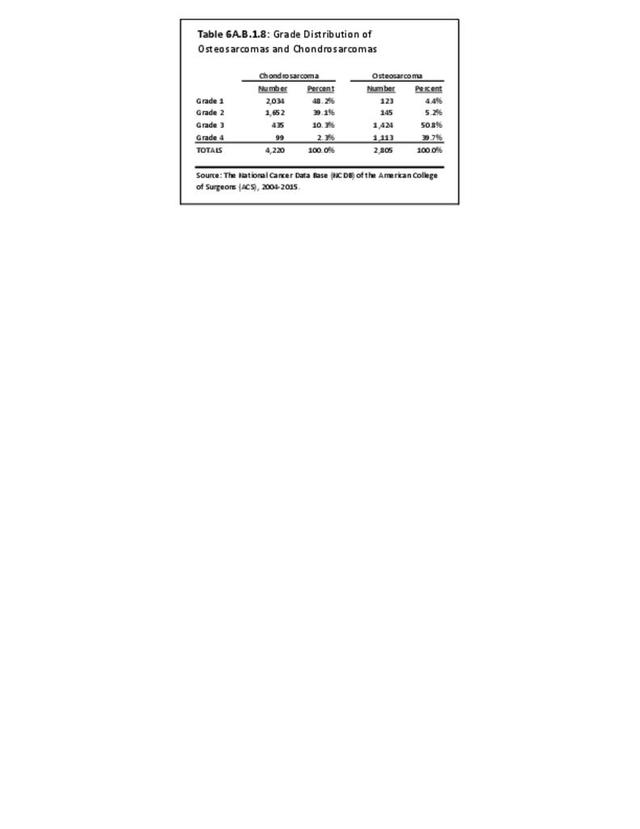
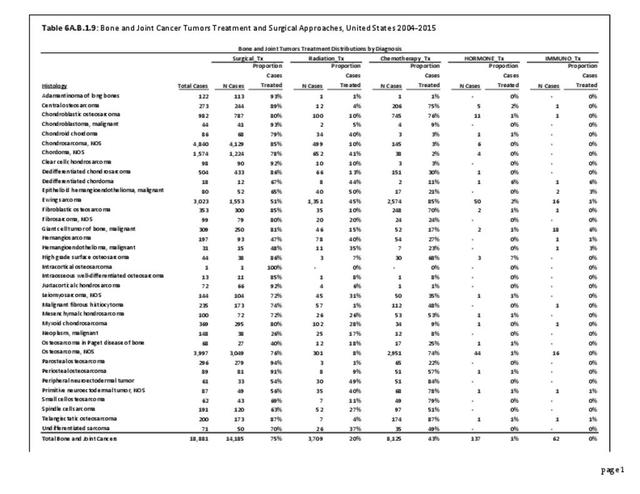
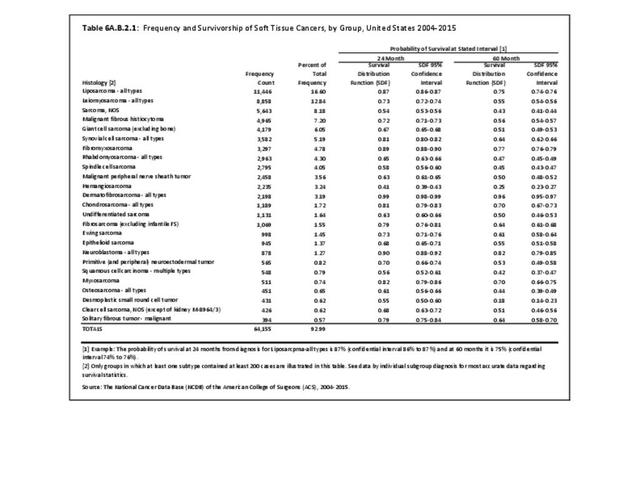
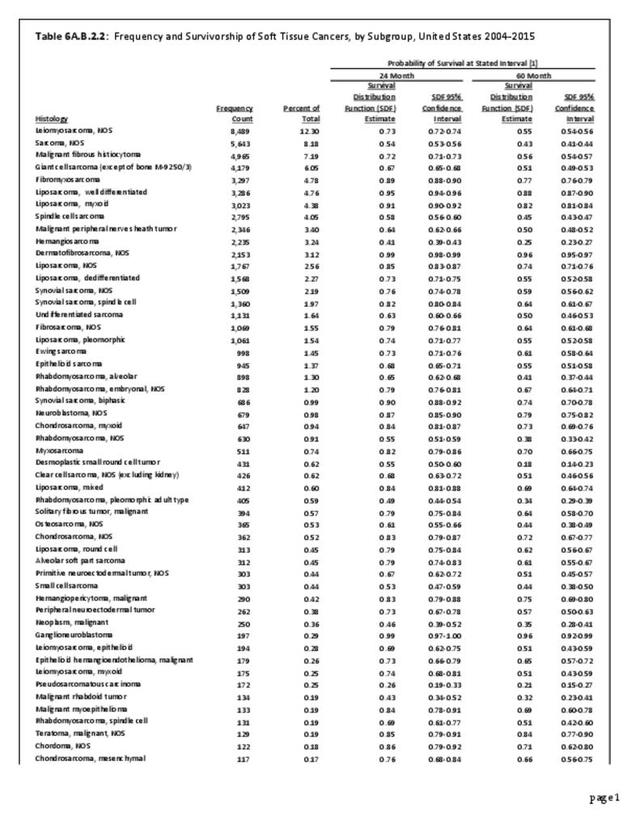
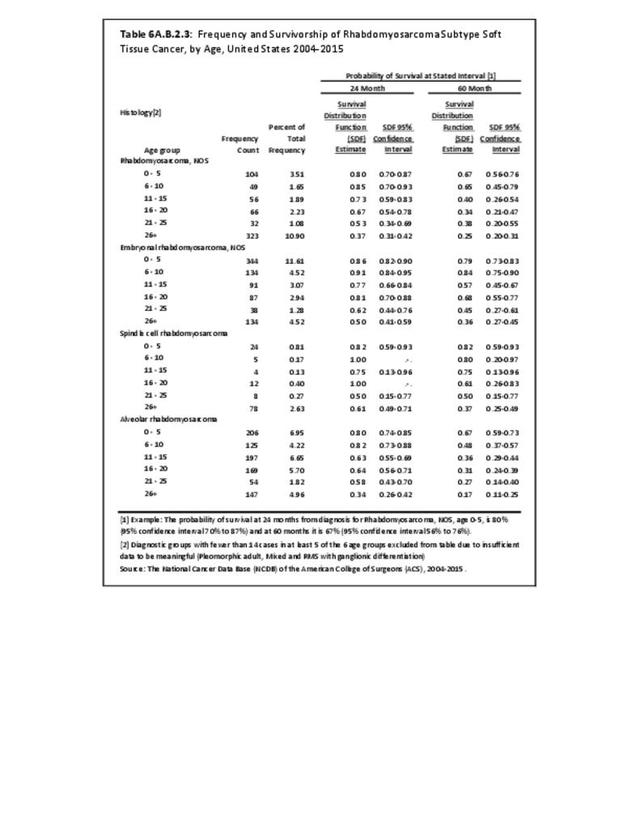
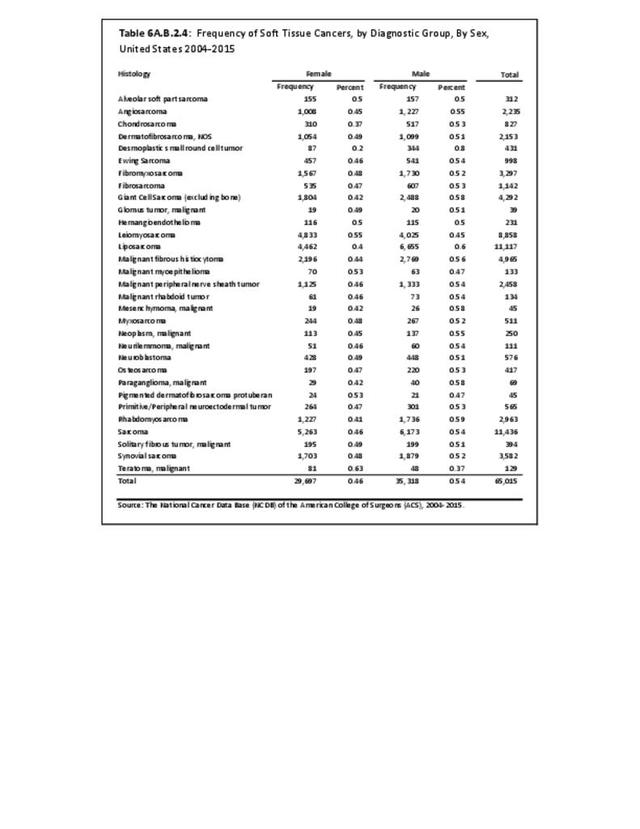
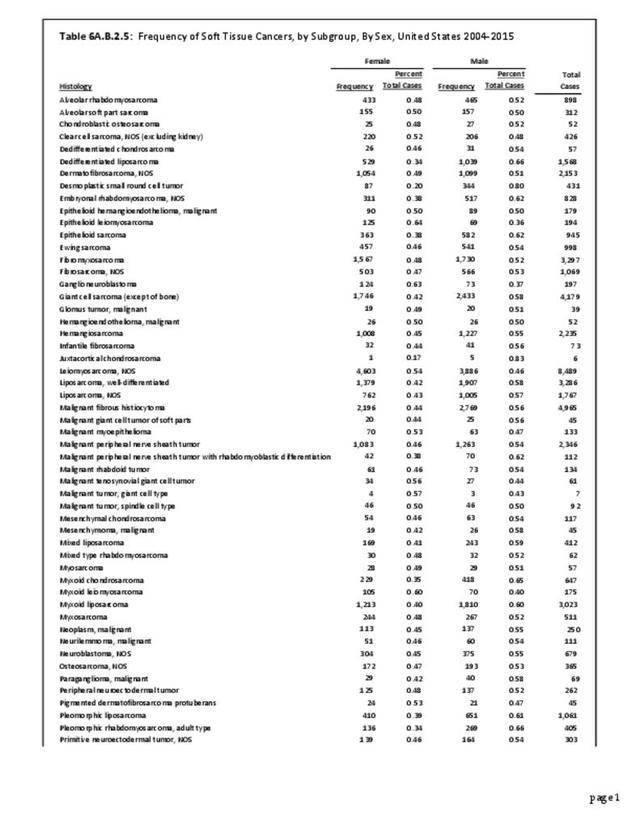
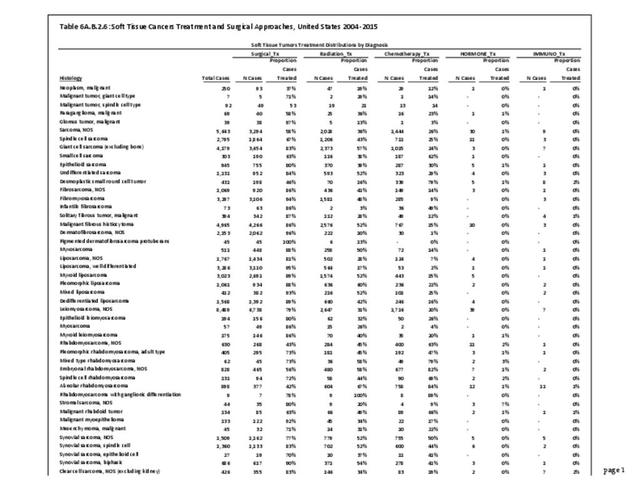
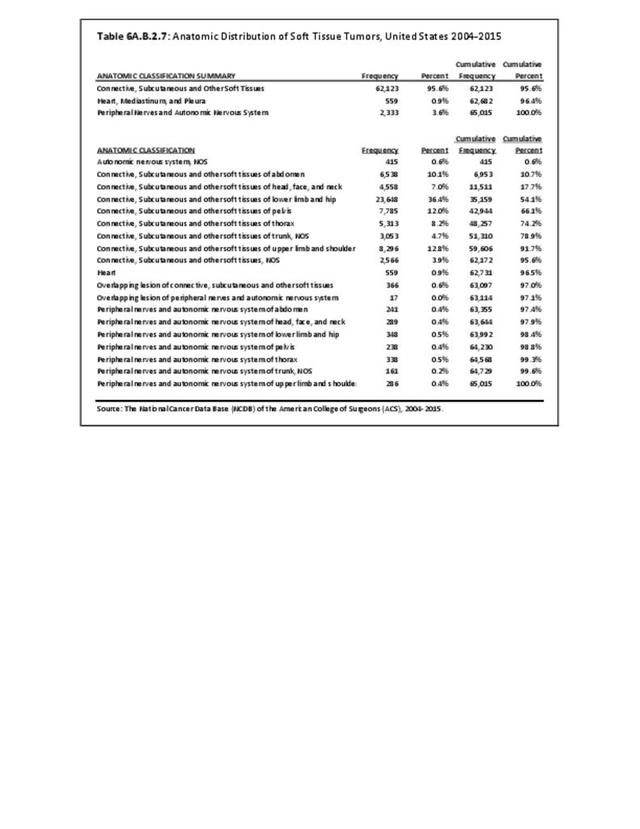
Musculoskeletal neoplasms cause significant morbidity and mortality, although less commonly than lung, breast, kidney, and certain other cancers. This significant burden is especially true in young patients who are more likely to develop cancers such as osteosarcoma, Ewing sarcoma, and rhabdomyosarcoma. Musculoskeletal neoplasms and sarcomas usually require concerted treatment efforts by coordinated medical teams. These teams are typically led by a subspecialty of physicians known as orthopedic oncologists. Because of the relative infrequency of musculoskeletal sarcomas, few institutions gather sufficient numbers to provide thorough epidemiologic and descriptive data. Therefore, tumor registry data are necessary to gather enough cases to generate meaningful data.
The following discussion is based on concerted analysis that incorporates the two largest tumor registries in the United States, the National Cancer Data Base (NCDB) of the American College of Surgeons (ACS) and the National Institutes of Health (NIH), National Cancer lnstitute’s Surveillance, Epidemiology and End Results (SEER) program. Actual incidence, death rates, and survival statistics are difficult to determine. The two databases derive slightly different numbers, and the numbers change annually with newly reported data. Thus, a direct comparison of NCDB and SEER is not possible. Sources for data cited in tables and graphs are shown. Sources available at the time of analysis may no longer be available.
The NCDB is a joint project of the Commission on Cancer of the American College of Surgeons and the American Cancer Society. The data used in this study and this report are derived from analysis of a cohort of patients registered and treated 2004-2015 and the resulting de-identified NCDB file comprising more than 1,500 Commission-accredited cancer programs. Data are collected from all institutions wishing to be accredited by the American College of Surgeons Commission on Cancer. Each accredited institution is required to report all patients with cancer treated at their institution, including annual follow-up data. Site visits and interaction between American College of Surgeons cancer database personnel and the local reporting institutions verifies a minimum of 90% case capture and reporting for each institution. Multiple internal checks verify the data accuracy. It is estimated that the approximately 1,500 reporting institutions each year treat approximately 72% of all patients with malignancies in the United States.1 The primary author was granted research access to the database under the Participant User File (PUF) research program. In his prior PUF-based research (including prior data reported in the predecessor of this publication), the accessible data was only for cases in patients 18 years old and older, thus creating age-associated limitations of the NCDB dataset. The most recent database reported herein, however, includes patients of all ages treated 2004-2015, inclusive. The American College of Surgeons and the Commission on Cancer have not verified and are not responsible for the analytic or statistical methodology employed or the conclusions drawn from these data by the investigator and authors of this chapter. Data from the NCDB was used in the analysis for certain demographic, treatment and survivorship analyses for musculoskeletal cancers.
The SEER database is the main program used by the National Cancer Institute (NCI) to support cancer surveillance activities. It is the most comprehensive and authoritative source of information on cancer incidence, prevalence, mortality, survival, and lifetime risk in the United States. The SEER Program currently collects and publishes cancer incidence and survival data from population-based cancer registries covering approximately 34% of the U.S. population.2 Data is available from 1974 to 2016 and includes more than 10 million cases.
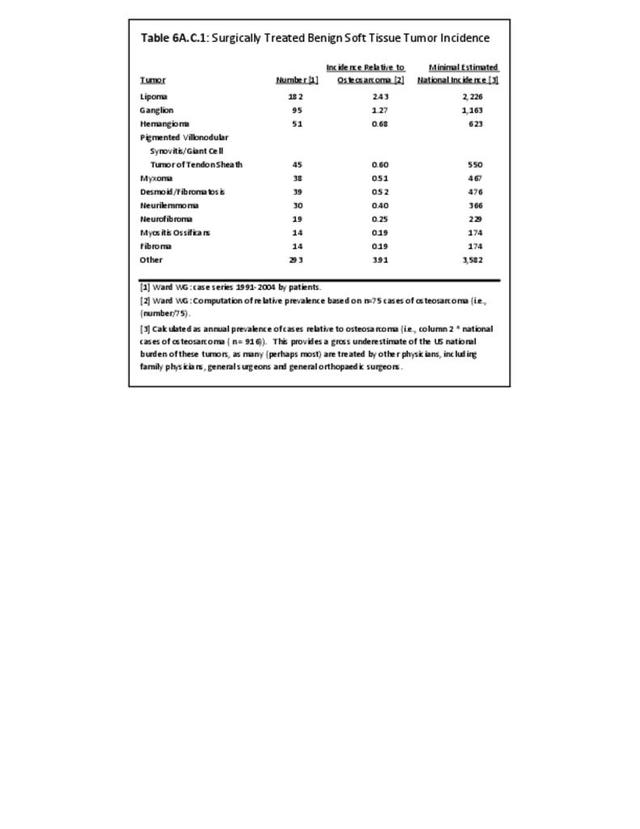
We also derived some tumor incidence estimates by analysis and extrapolation from one of the author's case series data compiled from his practice experience. Dr. William Ward was the only orthopedic oncologist at Wake Forest University Baptist Medical Center in Winston Salem, NC, during the period between 1991 and 2005. Virtually all cases of osteosarcoma in North Carolina were treated by one of the very few orthopedic oncologists in North Carolina during that time. Dr. Ward's personal surgical database contains detailed incidence data regarding many musculoskeletal neoplasms. Comparing the incidence of osteosarcomas in the United States with that treated by Dr. Ward, we were able to extrapolate, using similar proportional estimates, to the national incidence of tumors for which there were no national registry data. Typically, aggressive benign bone and benign soft tissue tumors are most likely to be treated by orthopedic oncologists rather than by non-oncological-trained surgeons. However, because there is no way to estimate the numbers of patients treated by orthopedic surgeons and other surgeons not specifically trained in orthopaedic oncology, the derived national data estimates will be conservative because of the methodology used. All estimates in this chapter derived from the above methodology will be clearly identified as an extrapolation via this incidence estimation.
Definitions
All tissues are made up of individual cells. Tumors, also known as neoplasms, are formed by uncontrolled and progressive excessive abnormal growth and multiplication of cells. In malignant tumors, the tumor cells continue to multiply and divide beyond the initial site. If unchecked, malignant tumors can cause death as they spread, or metastasize, to vital areas of the body. Benign tumors, on the other hand, remain localized and do not spread or metastasize to other body locations. They rarely threaten the life of the patient although they can cause significant injury or morbidity at the site of the tumor.
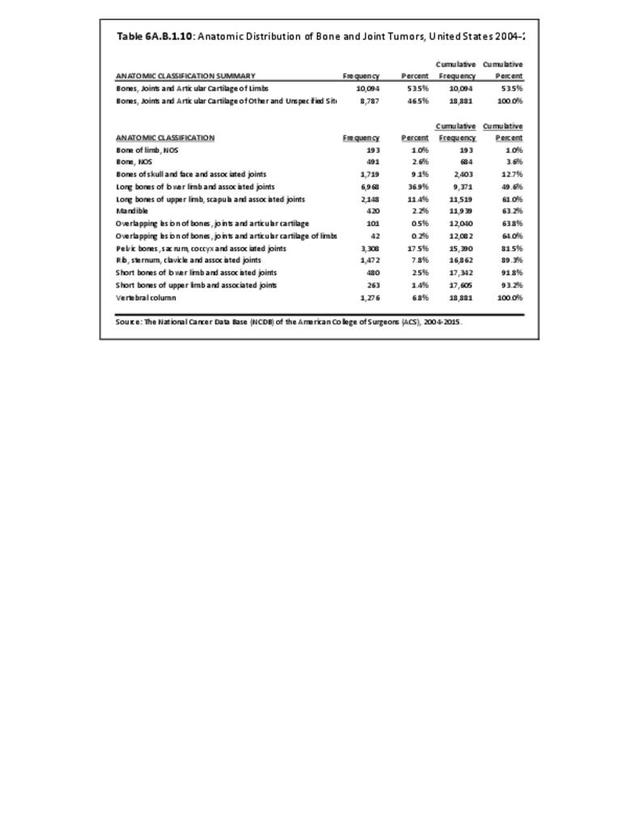
Muscle, bone, nerves, blood vessels, fat, and fibrous tissues are all connective tissues and are the tissue types that comprise musculoskeletal tissues and structures; therefore, tumors of these tissues form the basis of this report. Malignant tumors of the bone and connective tissue are also known as sarcomas, whereas cancers in most other organs are generally referred to as carcinomas.
Primary tumors are tumors of any organ or tissue that are composed of cells derived from that organ or tissue itself. Secondary, or metastatic tumors, are tumors in any organ or tissue that originated in a distant organ or tissue. Therefore, primary bone and soft tissue tumors originate in bone or connective tissue itself. Secondary or metastatic bone or connective tissue tumors began elsewhere and spread (metastasize) to the bone or connective tissues, retaining the cellular composition of the original tumor site. Primary tumors can be benign, which means they do not spread through the body to other sites, or malignant (cancerous), meaning they can and do spread to other places in the body.
Most musculoskeletal cancers, or sarcomas, are named by the Latin root word for the type of malignant tissue they produce. Thus, osteosarcomas are composed of malignant bone (osteo) producing cells; chondrosarcomas are composed of malignant cartilage (chondro) producing cells; liposarcomas are composed of malignant fat producing (lipo) cells; rhabdomyosarcomas create malignant muscle tissue (rhabdomyo); fibrosarcomas produce malignant connective tissue (fibro), and so on.
Secondary bone tumors that spread to the bone from malignancies in other organs such as lung, breast, and prostate cancers (metastatic cancers) are far more numerous than primary bone cancers. Although metastatic cancers to bone cause extensive morbidity from pain and fractures caused by bone weakening, such cancers are not the primary focus of this chapter. However, a section on secondary bone and joint cancers details some of the effects of this condition and its associated morbidity.
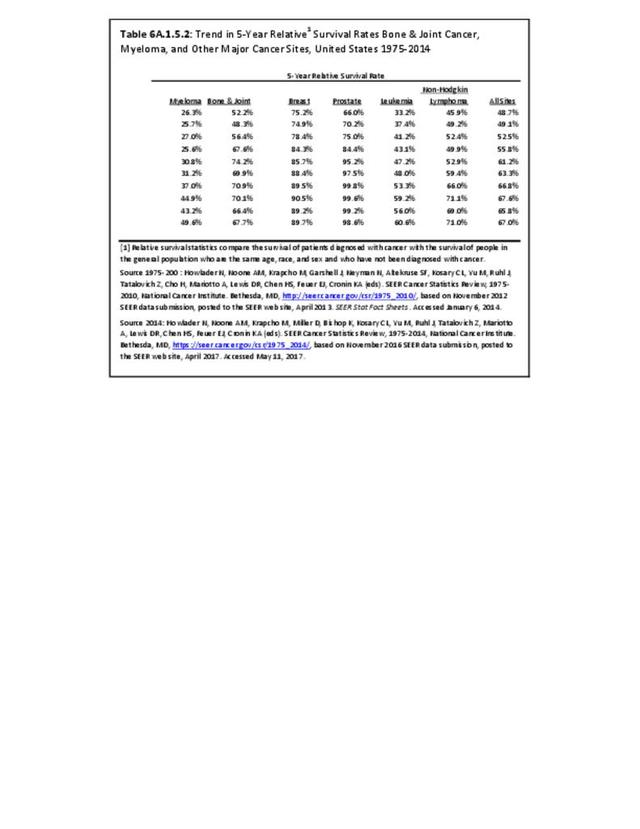
Hematologic (bone marrow) tumors are often not included in treatises on “bone cancer.” The three most common are leukemia (rarely weakens bone or causes fractures), lymphoma (can destroy bone structure and weaken bone causing fractures), and myeloma (often causes bone destruction). The latter two often cause bone weakness, pain, and fractures, so these two bone marrow tumors are included in most studies on bone cancers. Leukemias are rarely included since they only rarely cause significant bone weakness or fractures, therefore we have left them out of this chapter as well.
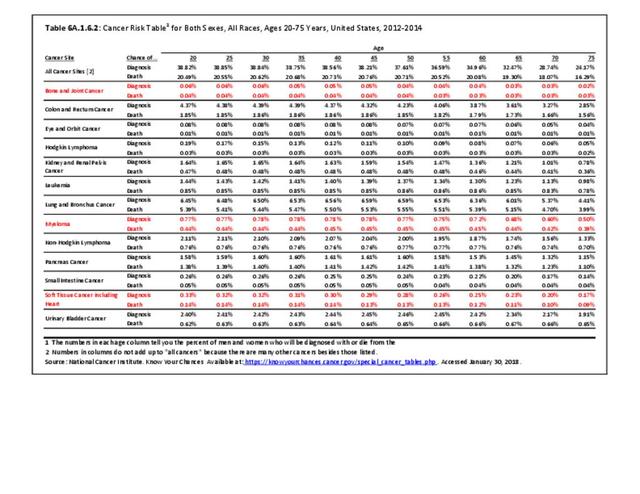
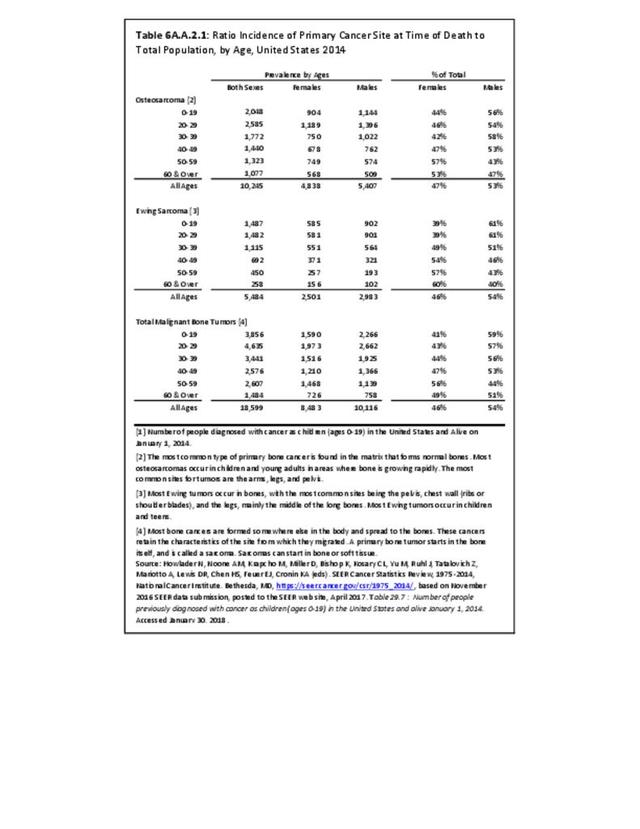
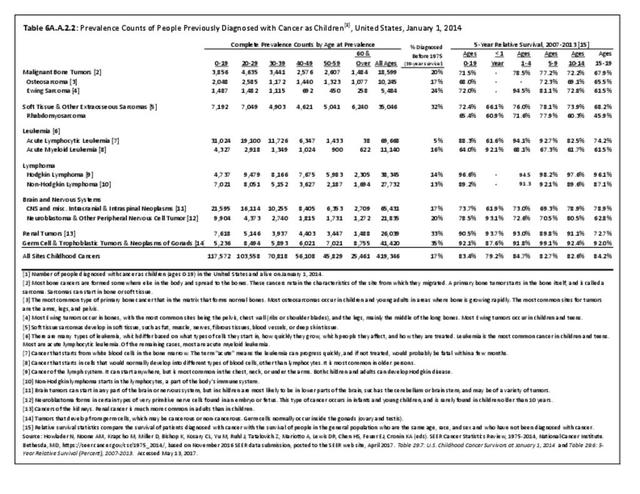
The incidence of cancer is defined as the number of new cancers of bone and connective tissue in a specific population during a year. The incidence rate is expressed as the number of cancers per 100,000 population at risk. In general, it does not include recurrences. Because of the low number of new cases, the incidence rate in this report is expressed as the number per one million population at risk.
Prevalence is defined as the number of people alive on a certain date in a population who have the disease and have previously had a diagnosis of the disease. It includes new (incidence) cases and pre- existing cases and is a function of past incidence and survival.
A cancer mortality rate is the number of deaths, with cancer as the underlying cause of death, occurring in a specific population during a year. It is calculated the same as the incidence rate.
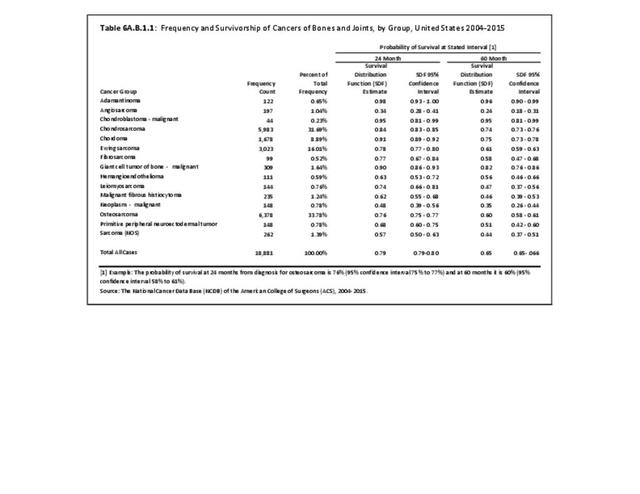
Cancer survival statistics are typically expressed as the proportion of patients alive at some point subsequent to the diagnosis of their cancer. Observed all-cause survival is an estimate of the probability of surviving all causes of death. Net cancer-specific survival (policy-based statistic) is the probability of surviving cancer in the absence of other causes of death. It is a measure that is not influenced by changes in mortality from other causes and, therefore, provides a useful measure for tracking survival across time, and comparisons between racial/ethnic groups or between registries. Crude probability of death (patient prognosis measure) is the probability of dying of cancer in the presence of other causes of death. It is a better measure to assess the impact of cancer diagnosis at an individual level since mortality from other causes play a key role. It measures mortality patterns experienced in a cohort of cancer patients on which many possible causes of death are acting simultaneously. The crude measure is reported as a cumulative probability of death from cancer rather than survival.3
Net cancer-specific survival measures are relative survival and cause-specific survival. Relative survival is the ratio of the proportion of observed survivors (all causes of death) in a cohort of cancer patients to the proportion of expected survivors in a comparable cohort of cancer-free individuals. The formulation is based on the assumption of independent competing causes of death. Cause-specific survival is a net survival measure representing survival of a specified cause of death in the absence of other causes of death. Estimates are calculated by specifying the cause of death. Individuals who die of causes other than those specified are considered to be censored.4
- 1. Mallin K, Browner A, Palis B, et al. Incident cases captured in the National Cancer Database compared with those in U.S. population based Central Cancer Registries in 2012-2014. Ann Surg Oncol 2019 Jun;26(6):1604-1612. doi: 10.1245/s10434-019-07213-1. Epub 2019 Feb 8. https://www.ncbi.nlm.nih.gov/pubmed/30737668 Accessed December 27, 2019.
- 2. National Cancer Institute, Division of Cancer Control & Population Sciences. Cancer Registry Resources: Surveillance, Epidemiology, and End Results (Seer) Program. https://epi.grants.cancer.gov/registries.html Accessed July 30, 2019.
- 3. National Cancer Institute, Division of Cancer Control & Population Sciences. Surveillance Research Program. Measures of Cancer Survival. Available at: https://surveillance.cancer.gov/survival/measures.html. Accessed January 5, 2018.
- 4. American College of Surgeons, National Cancer Database. About the National Cancer Database. https://www.facs.org/quality-programs/cancer/ncdb/about , accessed September 12, 2019.
Edition:
- Fourth Edition